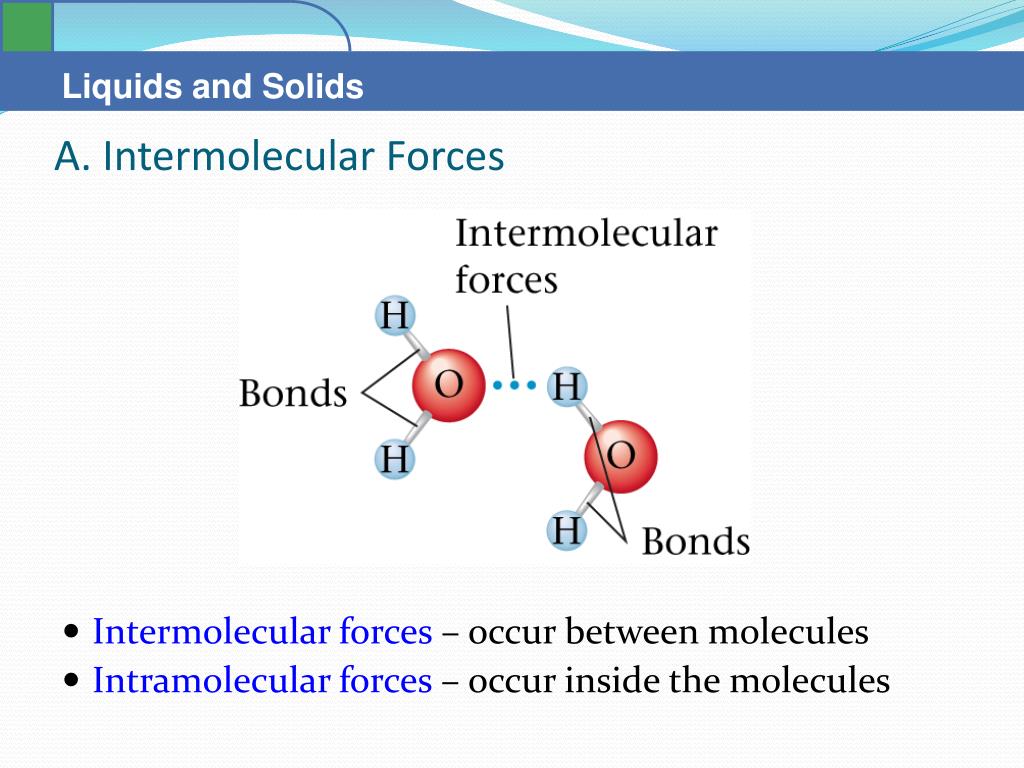
The H end has a small +ve charge and the Cl end has a small -ve charge. Polar covalent bonds exist as a dipole, e.g. Some people say that the shared electrons spend more time around one atom than the other atom, so this kind of covalent bond has a sort of ionic character. 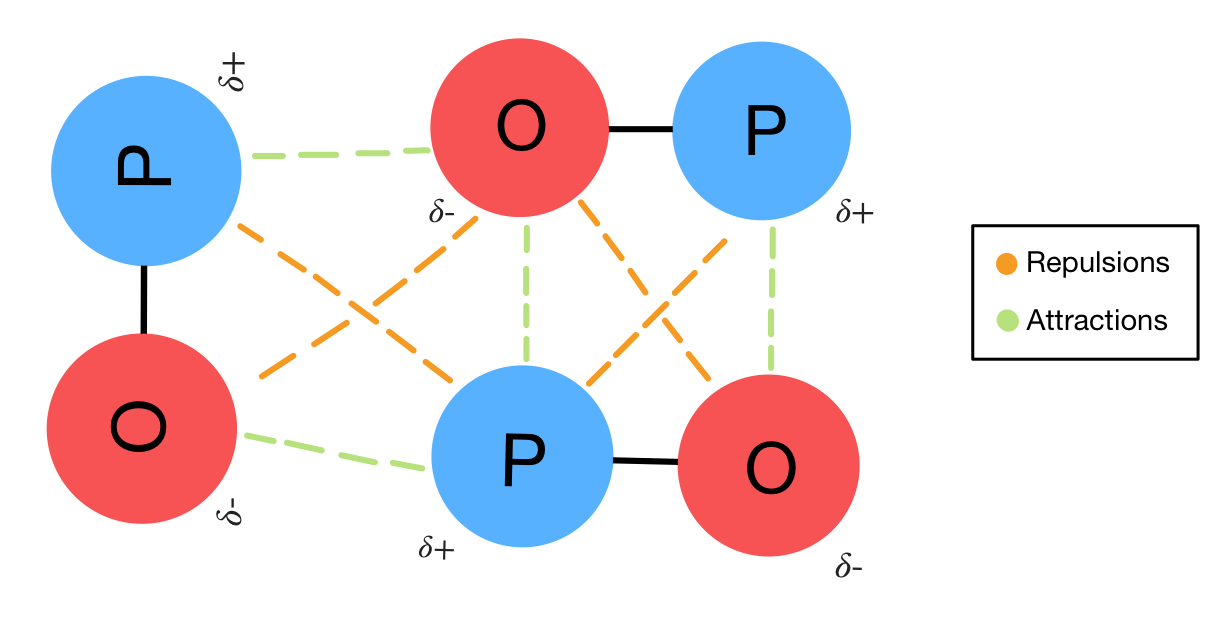 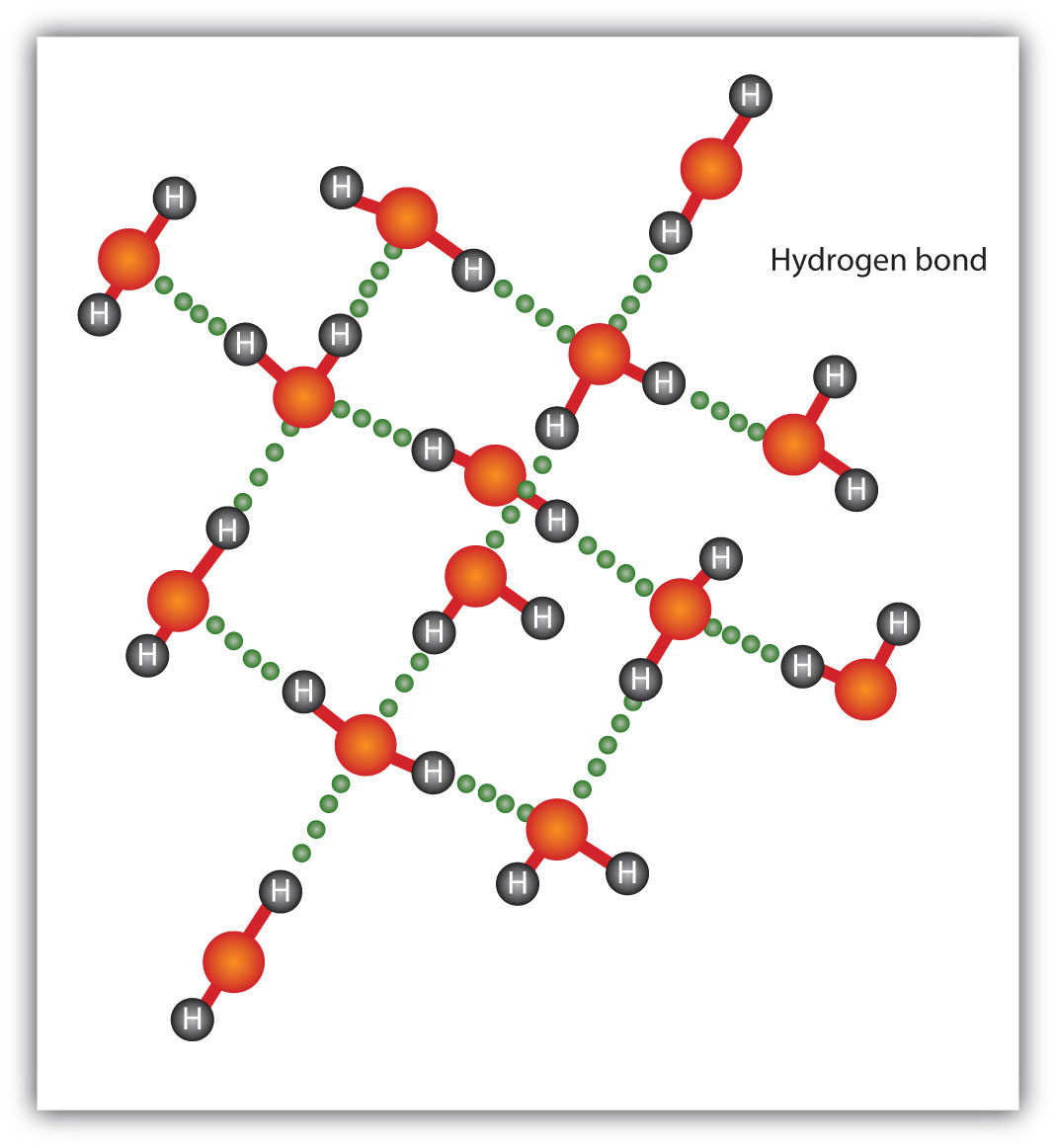
Polar covalent bonds form when atoms of two different elements each donate one electron to form a shared pair of electrons, but one atom has a greater share than the other atom. water, but are soluble in non-polar solvents, e.g. Non-polar covalent bonds (co-ordinate covalent bonds), in ammonium ion NH4 +, have electrons shared between the atoms and are not soluble in polar solvents, e.g. H: H.Įach atom donates one electron to form a pair of electrons that are equally shared by the two atoms. Non-polar covalent bonds exist between two or more, usually identical, atoms, e.g. Two or more atoms bound together by one or more covalent bonds form a molecule, with definite size, shape and arrangement of bonds.Īn atom or group of atoms covalently bound together may gain or lose one or more electrons to form ions.Ģ. Pairs of atoms may be bound together by the sharing of electrons between them in a covalent bond. Covalent bonds form by sharing of electrons. However, when molten or in solution, their electrical conductivity is high.ġ. Solids with ionic bonds have high melting points and low electrical conductivity. sodium chloride, NaCl, dissolves in water, the cations and anions separate, e.g. Ionic solids are usually transparent and have high melting points and boiling points. Ionic bonds are electrical links between atoms caused by the distribution of electrons around the nuclei of the bonded atoms. Ionic bonding occurs when positive and negative ions are held together in a crystal lattice by electrostatic forces. Ionic bonds, electrovalent bonds, form by transfer of electrons Chemical bonding is about atoms and electrons arranging into lower energy states.ġ1.01.1 Ionic bonds, electrovalent bonds, e.g. The three main types of bonding are ionic, covalent and metallic bonding.ģ. Chemical bonds are forces of attraction between the atoms together in a molecule or a crystal.Īll chemical bonds are caused by attractive force between positive and negative particles.Ģ. sodium chlorideġ1.01.9 Liquids with different viscosity, hydrogen bondsġ1.01.7 van de Waals forces (van de Waals bonds)Ģ0.0.13 van der Waals forces, Universal gas equationġ1.1.0 Chemical equations and ionic equations, conservation of massġ1.1.5 Burn steel wool and burn iron filingsġ1.1.1 Effervescent tablets, health salts, sodium bicarbonate, (baking soda)ġ1.2.1 Movement of ions, potassium permanganate solutionġ1.2.2 Movement of ions, sodium sulfate solutionġ1.2.3 Movement of copper ions in ammonium nitrate solutionġ1.2.4 Movement of suspended aluminium powderġ1.3.1 Colour change of diluted potassium permanganateġ1.3.4 Size of carbon atom in stearic acid moleculeģ.91 Size of particles and rate of reaction with balloonsġ1.6.1 Prepare salts by neutralization of a soluble acid and a baseġ1.6.2 Prepare salts by acids + metals or metal oxidesġ1.6.3 Prepare salts by acids + metal oxidesġ1.6.4 Prepare salts by acids + metal hydroxidesġ1.6.5 Prepare salts by acids + metal carbonatesġ1.6.6 Prepare salts by precipitation reactionsġ1.6.7 Prepare salts by direct union of elementsġ1.6.8 Prepare salts by bases + nonmetallic oxidesġ. Please send comments to: bonds, conservation of massġ1.4.1 Electric writing, sodium chloride with litmus paperġ1.2.0 Movement of ions and particles in solutionġ1.6.0 Prepare salts by different methodsġ1.01.1 Ionic bonds, electrovalent bonds, e.g.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
